Matter and Mixtures
1. Matter
Definition:
Matter is anything that has mass and occupies space. Everything around us—air, water, food, clothes, houses, and even our own bodies—is made up of matter.
Nature of Matter
Anything that occupies space and has mass is known as matter. Everything around us is a form of matter.
The huge buildings, bridges, electrons revolving around a nucleus, the DNA in our cells, the surrounding air, the land beneath our feet, etc. all matter.
A matter is said to be composed of particles which are basically atoms and molecules.
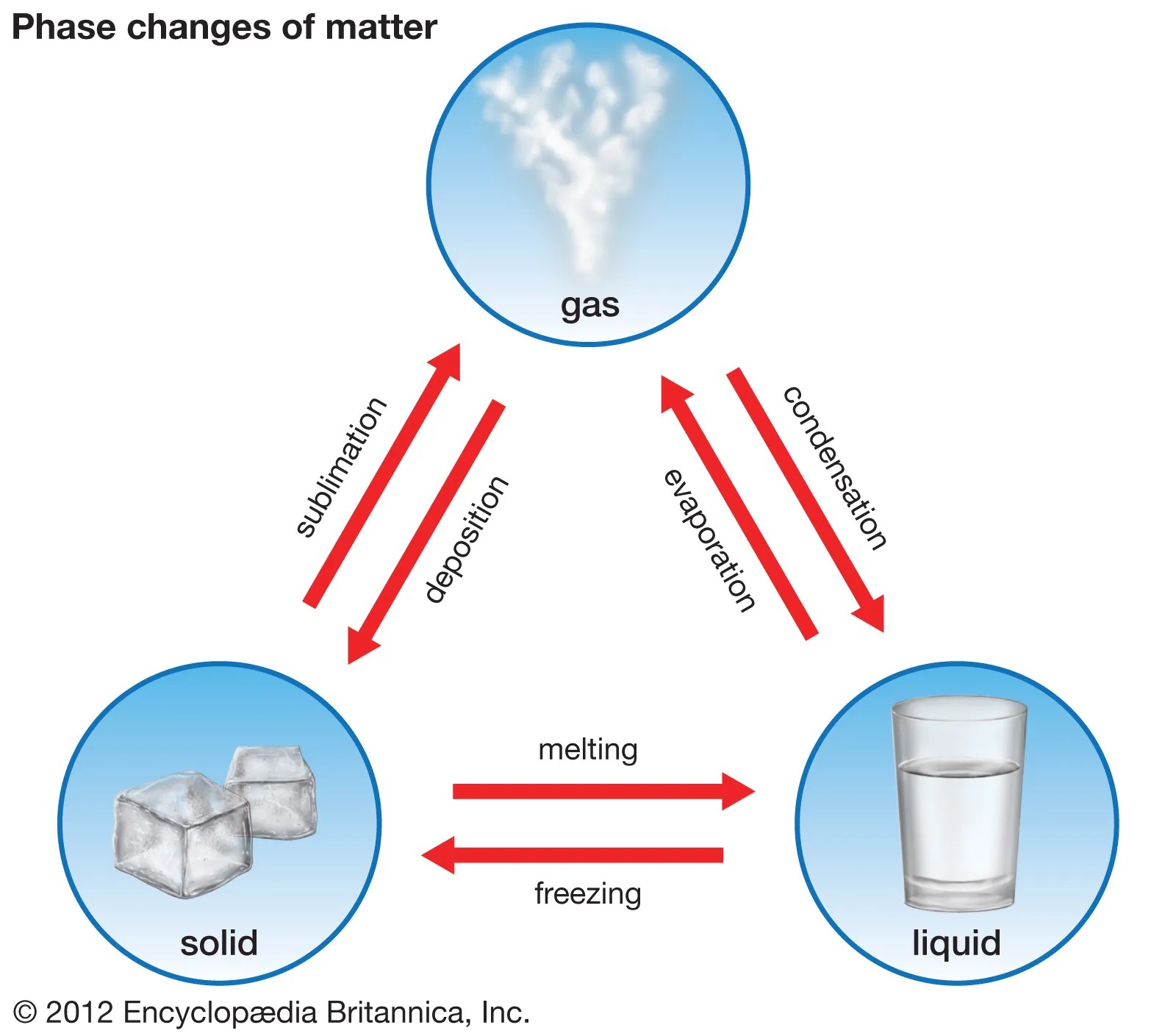
Depending upon its physical state, the nature of matter falls into three categories:
Solids:
The substances in which the particles are held close to each other with strong intermolecular forces are known as solids. The particles are strongly held at their positions and have only vibratory motion. Solids have a definite shape and definite volume. E.g.: Wood, iron, etc.
Liquids:
Those substances in which the intermolecular forces are weak enough to allow the movement of particles are known as liquids. The particles are held closely and have a higher degree of freedom than solids. Liquids have a definite volume but no definite shape; they generally take the shape of the container in which they are placed. E.g.: water, milk, etc.
Gases:
These types of matter have very weak forces between their molecules and hence the molecules are free to move. The distance between molecules is large as compared to solids and liquids. Gases have neither a fixed shape nor a definite volume. They tend to completely occupy the container in which they are placed. E.g. air, oxygen, hydrogen, methane, etc.
Summary: States of Matter
Solid – has a definite shape and volume.
Examples: rocks, books, tables.
Liquid – has a definite volume but takes the shape of its container.
Examples: water, juice, oil.
Gas – has no definite shape or volume; it spreads out to fill its container.
Examples: air, steam, oxygen.
Key Point: Matter can change form (solid, liquid, gas) through processes like melting, freezing, condensation, and evaporation.


2. Mixtures and Their Characteristics
What is a Mixture?
A mixture is a combination of two or more substances that are put together but do not form a new substance.
The materials in a mixture keep their own properties.
Mixtures can be separated through physical means.
Examples: halo-halo, trail mix, sand and gravel, seawater.
2.1 Homogeneous and Heterogeneous Mixtures
Homogeneous Mixtures (Uniform Mixtures)
The parts of the mixture look the same throughout.
The substances are evenly distributed.
Also called solutions.
Examples:
Salt dissolved in water
Vinegar
Sugar mixed in coffee
Heterogeneous Mixtures (Non-uniform Mixtures)
The parts of the mixture are not the same throughout.
You can often see or separate the different materials.
Examples:
Halo-halo
Mixed nuts
Sand and stones
2.2 Appearance and Uses
Uniform Mixtures (Homogeneous):
Appearance: Smooth, even, and the same throughout.
Uses: Beverages (juice, coffee), medicine (syrup), cleaning products (detergents).
Non-uniform Mixtures (Heterogeneous):
Appearance: Uneven; you can see different parts.
Uses: Salads, construction materials (gravel and cement), trail mix for snacks.
3. Separating Mixtures
3.1 Techniques in Separating Mixtures
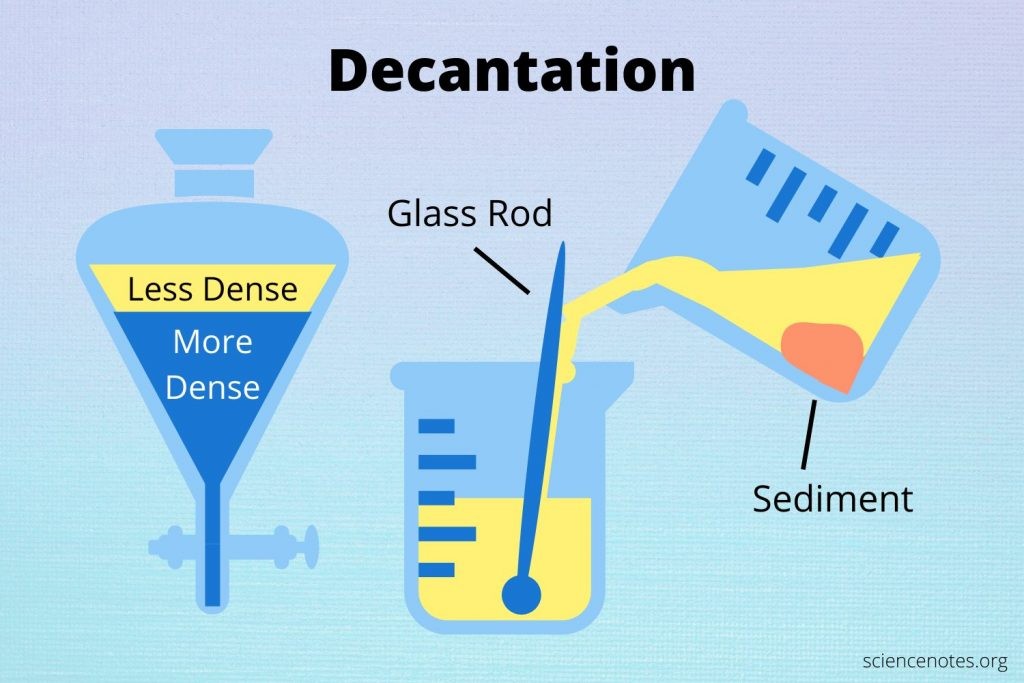
Decantation – gently pouring off liquid to separate it from solid particles that have settled at the bottom.
Example: Separating oil from vinegar, or water from rice after soaking.
Evaporation – heating a liquid until it turns into vapor, leaving the solid behind.
Example: Getting salt from seawater.
Filtration – using a filter to separate solid particles from a liquid or gas.
Example: Brewing coffee, straining calamansi juice to remove seeds.
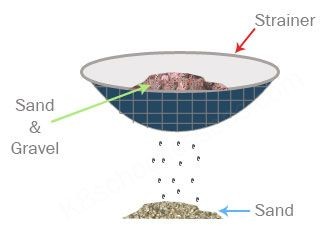
Sieving – using a sieve or strainer to separate larger particles from smaller ones.
Example: Sifting flour, separating sand from pebbles.
Using a Magnet – separating metallic substances from non-metallic ones.
Example: Removing iron nails from sand, separating scrap metals in junk shops.
3.2 Benefits of Separating Mixtures in the Community
Food Preparation: Clean rice by removing stones and husks.
Water Supply: Filter water to make it safe to drink.
Waste Management: Separate biodegradable and non-biodegradable wastes for recycling and composting.
Mining Industry: Extract valuable metals like iron and gold from soil.
Health and Sanitation: Purify salt and sugar for consumption.
Check Your Understanding
A. Recall Questions
What is matter? Give three examples.
Differentiate between homogeneous and heterogeneous mixtures.
What technique can you use to separate salt from seawater?
Why do we need to separate mixtures in our daily lives?
B. Critical Thinking
Imagine you are preparing halo-halo. How would you describe it as a mixture?
If your community has dirty river water, what separation techniques can be used to make it cleaner and safer to use? Explain your answer.
2. Mixtures and Their Characteristics
What is a Mixture?
A mixture is a combination of two or more substances that are put together but do not form a new substance.
The materials in a mixture keep their own properties.
Mixtures can be separated through physical means.
Examples: halo-halo, trail mix, sand and gravel, seawater.
2.1 Homogeneous Mixtures and
Heterogeneous Mixtures
Homogeneous Mixtures
(Uniform Mixtures)
The parts of the mixture look the same throughout.
The substances are evenly distributed.
They are also called solutions.
Examples:
Salt dissolved in water
Vinegar
Sugar mixed in coffee
Heterogeneous Mixtures
(Non-uniform Mixtures)
The parts of the mixture are not the same throughout.
You can often see or separate the different materials.
Examples:
Halo-halo
Mixed nuts
Sand and stones
2.2 Appearance and Uses
Uniform Mixtures (Homogeneous):
Appearance: Smooth, even, and the same throughout.
Uses: Beverages (juice, coffee), medicine (syrup), cleaning products (detergents).
Non-uniform Mixtures (Heterogeneous):
Appearance: Uneven; you can see different parts.
Uses: Salads, construction materials (gravel and cement), trail mix for snacks.
3. Separating Mixtures
3.1 Techniques in Separating Mixtures
Decantation – gently pouring off liquid to separate it from solid particles that have settled at the bottom.
Example: Separating oil from vinegar, or water from rice after soaking.
Evaporation – heating a liquid until it turns into vapor, leaving the solid behind.
Example: Getting salt from seawater.
Filtration – using a filter to separate solid particles from a liquid or gas.
Example: Brewing coffee, straining
calamansi juice to remove seeds.
Sieving – using a sieve or strainer to separate larger particles from smaller ones.
Example: Sifting flour, separating sand from pebbles.
Using a Magnet – separating metallic substances from non-metallic ones.
Example: Removing iron nails from sand, separating scrap metals in junk shops.
3.2 Benefits of Separating Mixtures in the Community
Food Preparation: Clean rice by removing stones and husks.
Water Supply: Filter water to make it safe to drink.
Waste Management: Separate biodegradable and non-biodegradable wastes for recycling and composting.
Mining Industry: Extract valuable metals like iron and gold from soil.
Health and Sanitation: Purify salt and sugar for consumption.
Check Your Understanding
A. Recall Questions
1. What is matter? Give three examples.
2. Differentiate between homogeneous
and heterogeneous mixtures.
3. What technique can you use to
separate salt from seawater?
4. Why do we need to separate mixtures
in our daily lives?
B. Critical Thinking
1. Imagine you are preparing halo-halo.
How would you describe it as a
mixture?
2. If your community has dirty river water,
what separation techniques can be
used to make it cleaner and safer to
use? Explain your answer.