Matter in Daily Life
What is Matter?
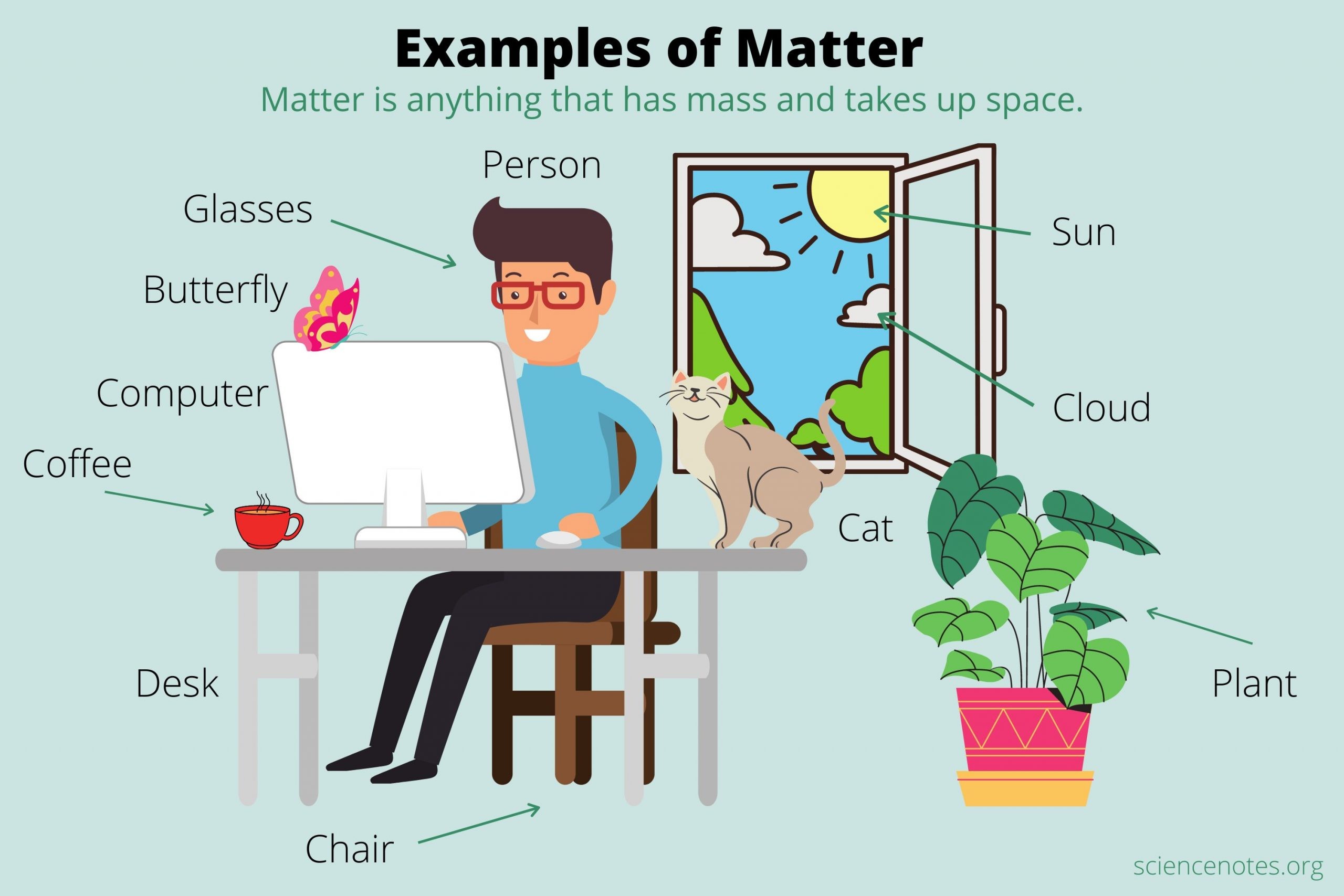
Matter is anything that has mass and takes up space. Everything you see, touch, smell, and even breathe is made of matter.
Whether it is the chair you are sitting on, the water you drink, or the air around you, all of these are matter.
Examples of Matter in Daily Life:
At home: your books, the food you eat, the water in your glass, and the air inside a balloon.
In the classroom: your pencil, your notebook, the floor tiles, and the chalk dust.
Outside: trees, rocks, clouds, and even the wind.
Matter can be small, like a grain of sand, or very big, like a mountain. No matter how big or small, if it has mass and occupies space, it is matter.
Key Idea: If you can weigh it and it occupies space, it is matter!
Matter and the Three States
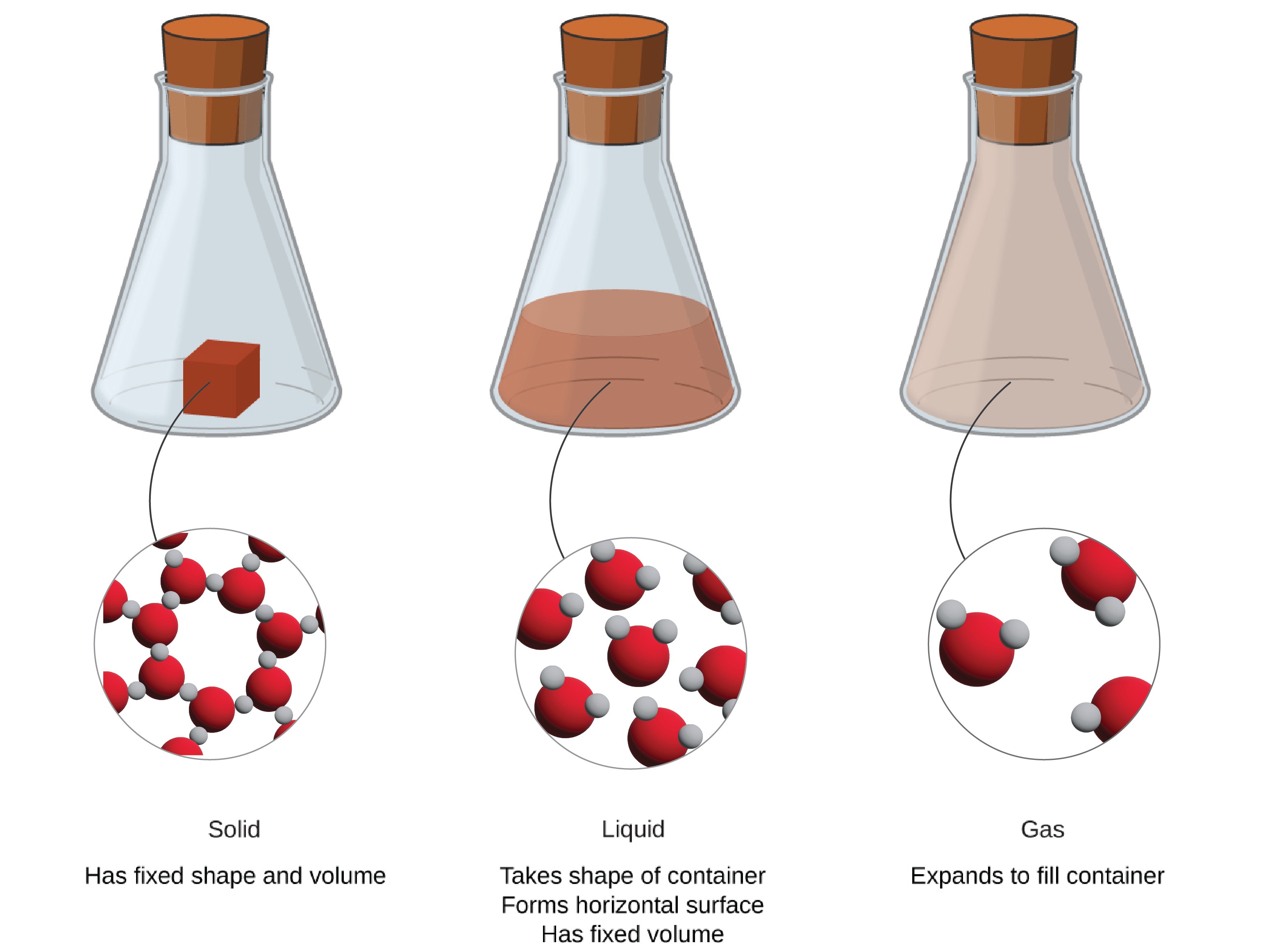
Matter exists in three common states: solids, liquids, and gases. Each has its own properties.
1. Solids
Shape: Definite (they keep their shape).
Volume: Definite (they keep their size).
Examples: a book, a stone, a chair.
If you place a rock on a table, it will not change shape or size on its own.
2. Liquids
Shape: No definite shape (they take the shape of their container).
Volume: Definite (they keep the same amount of space).
Examples: water, milk, juice.
A glass of water will change its shape depending on the container but still has the same volume.
3. Gases
Shape: No definite shape.
Volume: No definite volume (they expand to fill the container).
Examples: air, oxygen, helium in a balloon.
Air inside a balloon spreads out evenly, taking the shape and space of the balloon.
Measuring Matter
Solids are measured using balances and scales (mg, g, kg).
Liquids are measured using beakers, measuring cylinders, or bottles (mL, L).
Gases are harder to measure, but we can measure their mass with a balance and their temperature with a thermometer.
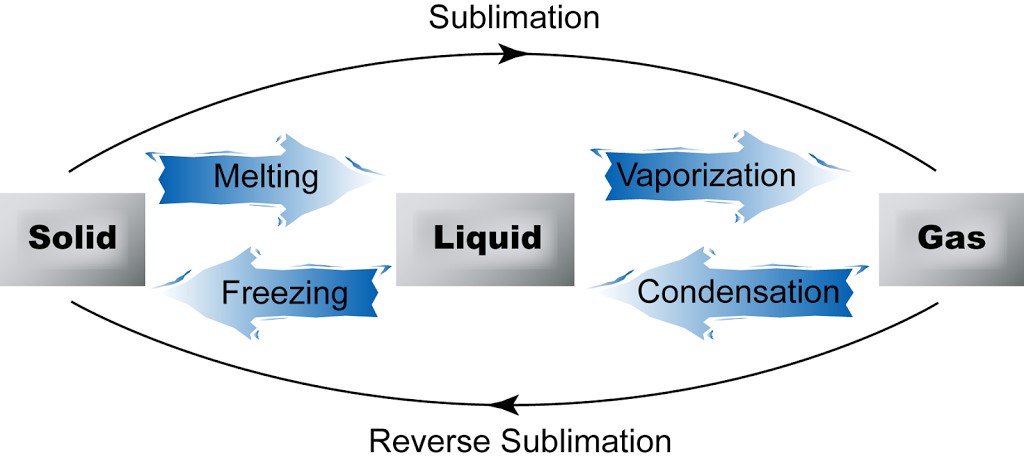
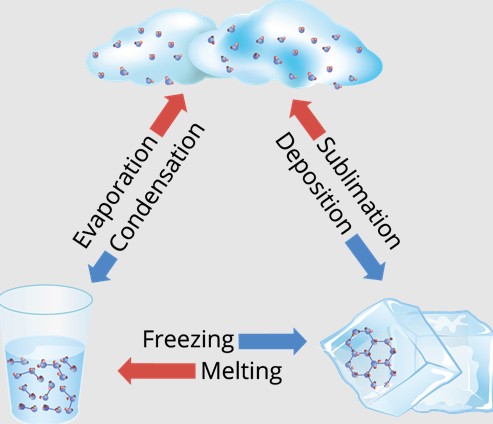
Changing States of Matter
Matter can change from one state to another when the temperature changes:
Solid to Liquid: melting (ice to water).
Liquid to Gas: evaporation/boiling (water to steam).
Gas to Liquid: condensation (water vapor to water droplets).
Liquid to Solid: freezing (water to ice).